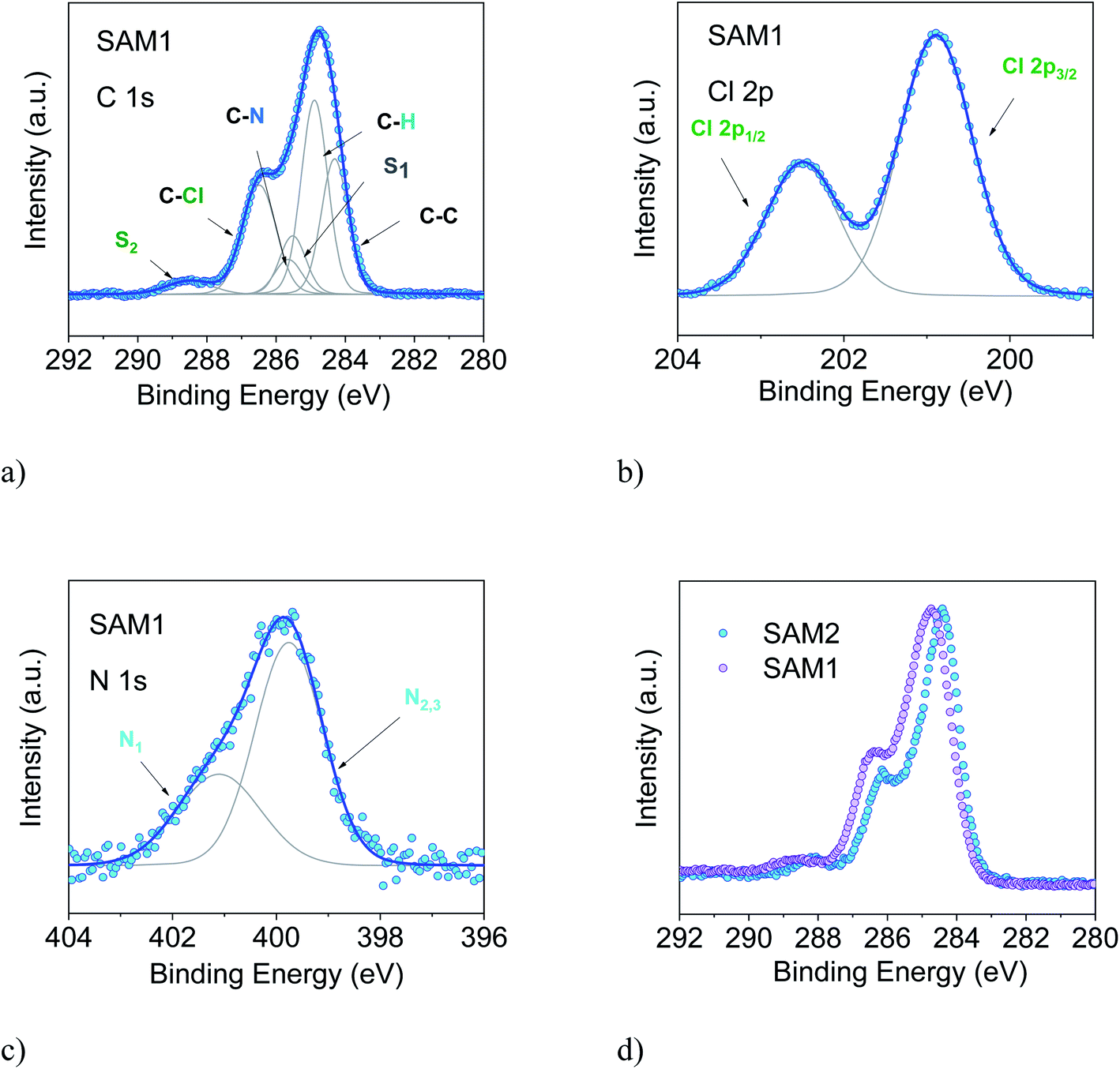
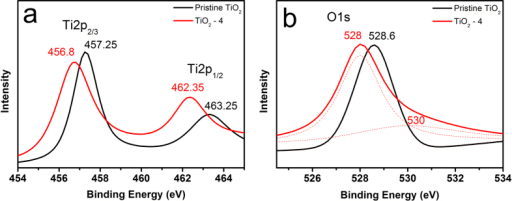
By XPS evidence is presented for the Ti 4+/Ti 3+ related redox catalysis and the details of the C and O-functionalities. For better comparison the data are normalized to each O1s peak maximum and separated vertically, but shown in the same scaling. A further proof of self-cleaning was the ability by the polyester-TiO 2 samples to inactivate again bacterial charge at the end of an inactivation cycle. Figure 3: XPS survey spectra (Mg K) of the PE-ALD layers deposited at 200 ☌ (red curve), 80 ☌ (blue) and rt (green) and the T-ALD at 200 ☌ (black). Evidence for the photocatalyst self-cleaning was found by XPS due to the lack of accumulation of bacterial residues on the polyester-TiO 2. Peak shapes: single peaks, doublets, and extra complexity. After 30 min RF-plasma pretreatment of the polyester samples, the fastest bacterial inactivation was observed concomitant with (a) the largest ratio of surface oxidized to the reduced functionalities as determined by XPS, (b) a strong sample optical absorption as seen by DRS and (c) the highest concentration surface OH-radicals monitoring the fluorescence of the hydroxy-terephthalic acid. In this advanced example, the scientist, for ease of viewing, has also ignored the 2p3/2 and 2p1/2 of the SiO2 peaks. The drawing shows the relation between the Silicon atoms and their XPS peaks. Here is an example of Si (2p) spectrum from a Synchrotron facility. Figure 1: C 1s region measured using a Kratos Axis 165 from a nylon sample. Silicon peak-fitting is can be very challenging.
Evidence presented for the RF pretreatment of polyester enhances the TiO 2 coating generation of oxidative species/radicals under a low level actinic light irradiation. The C 1s spectrum clearly contains two chemically shifted C 1s peaks however the more subtle shifts associated with the peaks labelled CH2 are the reason peak fitting is an important tool in XPS.
